New study shows the structure of transport protein involved in human fat metabolism
Together with his research group, Pontus Gourdon has determined the molecular structure of a human protein that is a key regulator for how fat is removed from fat cells. These findings open up for the development of new treatments of obesity and diabetes.
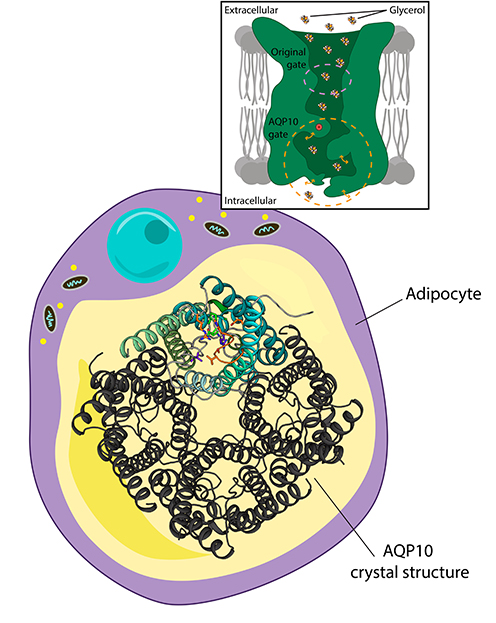
A new study by Associate Professor Pontus Gourdon's research group has determined that human AQP10 is a key regulator in the transportation of the lipolysis-derived glycerol moiety of fat from fat cells, and furthermore revealed the molecular structure of this protein. Thereby, they have identified how the glycerol transport is regulated by an unexpected “molecular gate".
Together with collaborators, Kamil Gotfryd, Julie Winkel Missel, Kaituo Wang and Pontus Gourdon from the Membrane Protein Structural Biology group have conducted the study, and it is, to their knowledge, the first structure of a human membrane protein ever determined in Denmark. “Our findings represent basic science and provide an understanding of the determinants that govern the important transport of glycerol in health and disease. Additional efforts are required for this to be exploited in translational applications" says research group leader Pontus Gourdon.
The goal is that the new findings will help to identify new treatments against for example diabetes and obesity.
Their results are described in the paper entitled "Human adipose glycerol flux is regulated by a pH gate in AQP10" that is published in the prestigious journal Nature Communications today.
